TOWARDS TRUE SSTABILIZATION
ENERGIES OF H-BONDED AND STACKED DNA BASE PAIRS
Petr
Jurečka and Pavel Hobza
Research
Center for Complex Molecular Systems and Biomolecules,
Jaroslav
Heyrovský
Institute of Physical Chemistry, Academy of Sciences of the Czech Republic,
18223 Praha
The
study of binding energies of Adenine...Thymine and Guanine...Cytosine base
pairs in vacuo was aimed to get an information on relative order of different
structure motives on the energetical scale with special focuse on differences
between H-bonded and stacked structures. We also aimed to find a general way to
reliable interaction energies of the weakly bonded complexes.
Structures
taken from previous MD/quench studies were fully optimized at RI-MP2 [1,2]
level with TZVPP [5s3p2d1f]/[3s2p1d] basis set. To approach complete basis set
(CBS) limit, convergency of both HF energy and MP2 correlation energy was
studied employing augmented correlation-consistent basis sets
aug-cc-pV(D,T,Q)Z. Is was found that molecular interaction energies close to
the CBS limit may be obtained by 2-point extrapolation [3] using aug-cc-pVDZ
and aug-cc-pVTZ basis sets.
To
account for higher order correlation effects convergency of CCSD(T)→MP2
correction term (ΔEcorrMP2 - ΔEcorrCCSD(T))
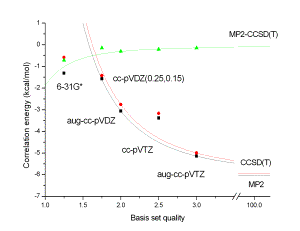
was investigated. For the formamide….formamidine complex (Fig. 1) which is a
model for adenine..thymine interaction MP2 and CCSD(T) correlation interaction
energies were evaluated with various basis sets up to aug-cc-pVTZ (Fig. 2). It
was shown that unlike the correlation energy itself the CCSD(T)→MP2
difference is almost basis set independent. Rather accurate values were
obtained with relatively small 6-31G*(0.25) and cc-pVDZ(0.25,0.15) basis sets.
Because the latter one performs well also for stacked complexes [4] it can be
recommended for evaluation of the Δ term of extended complexes possessing
both H-bonded and stacked structures.
Interaction
energies of the DNA base pairs obtained by combination of the CBS
extrapolations of MP2 interaction energies and the CCSD(T)→MP2 correction
are in good agreement with experiment.
Fig. 1 Fig.2
References
1)
M. Fayereisen, G. Fitzgerald & A. Komornicki, Chem. Phys. Lett., 208
(1993), 359.
2)
Ahlrichs R., Bär M.& Häser M., Chem. Phys. Lett., 162 (1989), 165.
3)
A. Halkier, T. Helgaker & P. Jørgensen, Chem. Phys. Lett.,
302 (1999), 437-446.
4)
Hobza P. & Šponer J., Chem. Phys. Lett., 288 (1998), 7-14.