Fluorescent proteins as molecular antennas: structure and directionality
J. Lazar1,2, J. Myšková1, O. Rybakova1,2, J. Brynda1,3
1Inst. of Organic Chemistry and Biochemistry CAS, Flemingovo nám. 2, 16000 Prague, Czech Rep.
2Inst. of Microbiology CAS, Zámek 136, 37333 Nové Hrady, Czech Rep.
2Inst. of Molecular Genetics CAS, Vídeňská 1083, 14220 Prague, Czech Rep.
lazar@uochb.cas.cz
The most important fluorescent molecules in biology are fluorescent proteins[1]. Despite their widespread use, it is largely unappreciated that they do not behave as point sources of light, but as antennas. The rate at which fluorescent proteins absorb light depends on their orientation with respect to the incoming light wave[2], and the emission of light by fluorescent proteins is directional. Directionality of optical properties of molecules is described by a vector, the transition dipole moment (TDM). Here we present the results of our optical measurements of FP crystals that, when combined with knowledge of the crystal structures, allowed us to determine the molecular orientations of absorption TDMs in several representative fluorescent proteins. Knowing the TDM orientations will allow quantitative interpretations of fluorescence resonance energy transfer experiments, determinations of molecular orientations by polarization-resolved microscopy, rational development of molecular probes, and other novel applications of these exceedingly useful molecules.
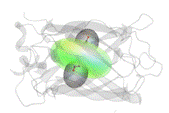
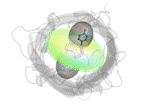
Figure 1. Structure of the molecule of the enhanced green fluorescent protein (eGFP), with the directionality of its light absorption and emission indicated
1. M. Chalfie, Y. Tu, G. Euskirchen, W. W. Ward, D. C. Prasher, Green fluorescent protein as a marker for gene expression. Science 263, 802-805 (1994).
2. S. Inoue, O. Shimomura, M. Goda, M. Shribak, P. T. Tran, Fluorescence polarization of green fluorescence protein. Proc Natl Acad Sci U S A 99, 4272-4277 (2002).
The work was supported by the European Regional Development Fund; OP RDE; Project: "ChemBioD- rug" (No. CZ.02.1.01/0.0/0.0/16_019/0000729