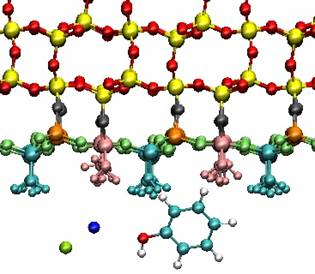
Behavior of water, ions and small organic
molecules near quartz (101) surfaces
O. Kroutil1, Z. Chval1,
H. Barvíková2 and M. Předota2
1 Faculty of Health and Social
Studies,
2
Faculty of Science, University of South Bohemia, Branišovská
31, 370 05 České Budějovice,
Czech Republic
* email: okroutil@gmail.com
We focused our attention on molecular
dynamics simulations of the interactions of quartz surfaces with aqueous solutions
of ions and small organic molecules representing functional groups of organic
matter.
Simulated quartz surfaces covered the range
of surface charge densities 0.00, -0.03, -0.06 and -0.12 C/m2,
approximately corresponding to pH values 4.5, 8.5, 9.5 and 10.
We found increasing water ordering within
two water layers closest to the surface with increasing surface charge density.
Also sodium ions adsorption increase with increasing charge density and
specific adsorption pattern can be found between negatively charged Si-O-
species and sodium cations.
Humic acids and humates have heterogeneous and
complex structures with different functional groups including acidic, hydrophilic,
and hydrophobic groups. Humic acids are able to
interact with both organic and inorganic substances such as nutrients, metals, hydrophobic
organic compounds and mineral surfaces. Understanding their structure and interactions
can give us important information about their degradability, toxicity, and
transport properties. Also they are one of the major causes of so-called
‘bio-fouling’ of nanofiltration and reverse osmosis
membranes which are used in industry for water purification, water desalination
and wastewater treatment.
Adsorption of small organic molecules
representing basic building blocks of larger biomolecules
and organic matter is also subject of this study. As model molecules, benzoic
acid, phenol, and salicylic acid were chosen.
We gratefully acknowledge support
from the Grant Agency of the Czech Republic P208/12/0622 and the Ministry of
Education, Youth and Sports of
the Czech Republic ME09062.