Intramolecular interactions in ferrocene phosphinostibines from the perspective of structural analysis and computational methods
D. Rezazgui, J. Schulz, P. Štěpnička
Charles University, Faculty of Science, Hlavova 8, 128 00, Praha 2, Czech Republic.
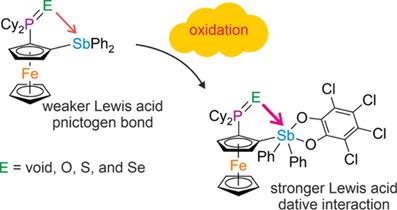
This contribution examines intramolecular non-covalent interactions in ferrocene phosphinostibines from the perspective of X-ray structural analysis and computational methods. Hybrid ligands combining phosphine and stibine donor groups were prepared in both flexible and rigid ferrocenyl frameworks, including 1,2-disubstituted phosphinostibines, their phosphine chalcogenides, gold complexes, and the corresponding oxidized stiboranes.
The primary focus is placed on intramolecular donor-acceptor interactions studied via single-crystal X-ray diffraction and theoretical calculations. The diffraction data reveal a clear dependence of the intramolecular contacts on the electronic state of the antimony center. Whereas neutral phosphinostibines show only limited evidence for attractive donor-acceptor interaction, oxidation to stiboranes generates significantly shortened P···Sb, Au···Sb and E···Sb contacts (E = O, S, Se), indicating pronounced pnictogen-centered Lewis acidity and effective intramolecular donor stabilization. In the rigid 1,2-disubstituted ferrocene series, these effects are particularly well resolved, allowing direct comparison of the structural consequences of donor type, oxidation state, and geometric preorganization. The shortest and most distinct interactions are observed for oxygen donors, while the sulfide and selenide analogues exhibit weaker but still recognizable trends.
The nature of these non-covalent interactions was further investigated using Density Functional Theory (DFT) and Intrinsic Bond Orbital (IBO) analysis together with Atoms In Molecules (AIM) analysis. The calculations confirmed the presence of pnictogen bond which upon oxidation of the stibine moiety to stiborane changed into covalent interaction [1].
The synergy between X-ray diffraction and quantum chemical modeling thus enables the rational design of new ligands, where these secondary interactions can be utilized to fine-tune reactivity.
This work was supported by the Czech Science Foundation (project no. 21-02316S) and Charles University Research Centre program (project no. UNCE/24/SCI/010). Computational resources were provided by the e-INFRA CZ project (ID: 90254), supported by the Ministry of Education, Youth and Sports of the Czech Republic.
[1] a) D. Rezazgui, J. Schulz, P. Štěpnička, Chem. Commun. 61 (2025) 18705-18708. b) D. Rezazgui, J. Schulz, P. Štěpnička, Inorg. Chem. 64 (2025) 11075-11092. c) J. Schulz, J. Antala, D. Rezazgui, I. Císařová, P. Štěpnička, Inorg. Chem. 62 (2023) 14028-14043.