Polymorphism of Helicenes studied by DSC and VT-XRD
V. Pokorný1, J. Obuch1, Š. Kocian2, V. Štejfa2
1Institute of Macromolecular Chemistry, Czech Academy of Sciences, Heyrovského nám. 2, 162 00, Prague 6, Czech Republic
2 Department of Physical Chemistry, University of Chemistry and Technology, Prague, Technická 5, 166 28 Prague 6, Czech Republic
pokorny@imc.cas.cz
Helicenes are a class of inherently chiral, π-conjugated polycyclic aromatic hydrocarbons characterized by their helical structures and unique chiroptical properties [1]. Their rigid yet non-planar geometry makes them valuable model systems for investigating the relationship between molecular shape and crystal packing [2]. In the solid state, subtle variations in intermolecular interactions can lead to polymorphism, which can significantly influence the physical properties of these materials. Despite extensive crystallographic studies of helicenes [3], polymorphism in shorter homologues such as [4]- and [5]-helicene remains insufficiently explored.
New polymorphs of [4]-helicene and [5]-helicene were identified by differential scanning calorimetry (DSC, TA Q2000) and characterized by variable-temperature powder X-ray diffraction (VT-PXRD; Anton Paar XRDynamic 500 equipped with a TTK600 temperature chamber, rotating 0.5 mm capillary). Temperature-dependent PXRD measurements were used to monitor phase transitions and isolate the new forms. Crystal structures were determined from powder data by indexing and structure solution using FOX, followed by Rietveld refinement in Jana2020.
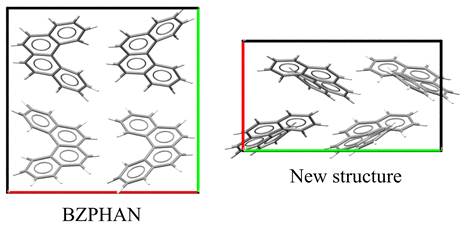
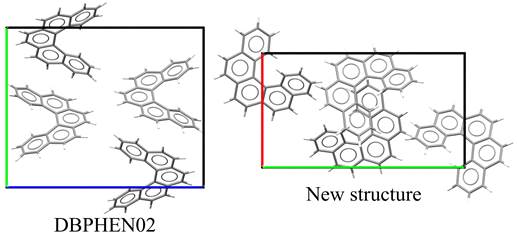
An overview of previously reported and newly identified polymorphs is given in Table 1. While the molecular conformation remains unchanged across all structures, significant differences are observed in crystal packing (see Figures 1 and 2). These packing variations correlate with differences in melting temperature, suggesting changes in lattice stability between polymorphs.
Table 1. Parameters of previously known and newly determined structures of [4]-helicene and [5]-helicene.a
|
polymorphb |
a / Å |
b / Å |
c / Å |
β / ˚ |
Z |
Space group |
Tmelt / ˚C |
|
[4]-helicene |
|||||||
|
BZPHAN |
14.660 |
14.157 |
5.785 |
90 |
4 |
P212121 |
66 |
|
New |
14.497 |
10.400 |
8.025 |
90 |
4 |
Pbc21 |
45 |
|
[5]-helicene |
|||||||
|
DBPHEN02 |
5.815 |
14.178 |
17.498 |
94.41 |
4 |
P21/c |
155 |
|
DBPHEN03 |
26.013 |
8.898 |
19.494 |
103.94 |
12 |
A2/a |
178 |
|
DBPHEN05 |
20.307 |
8.977 |
7.756 |
90 |
4 |
Pbcn |
n.d.c |
|
New |
9.924 |
17.536 |
8.395 |
90 |
4 |
Pna21 |
162 |
a parameters α and γ are 90˚ in all cases. β = 90˚ indicates an orthorhombic cell.
b identified by the CSD refcode, if available.
c n.d. stands for not determined.
|
|
|
Figure 1. Comparison of crystal packing in different polymorphs of [4]-helicene. |
Figure 2. Comparison of crystal packing in different polymorphs of [5]-helicene. |
These findings extend the known structural diversity of helicenes and support their use as model systems for packing-driven polymorphism in rigid π-conjugated molecules.
1. M. Cei, L. Di Bari, F. Zinna, Chirality, 35, (2003), pp. 192-210
2. Y. Shen, C. Shen, Chem. Rev., 112, (2012), pp. 1463-1535
3. J. A. Schmidt, E. H. Wolpert, G. M. Sparrow, E. R. Johnson, K. E. Jelfs, Cryst. Growth Des., 23, (2023), pp. 8909-8917
We acknowledge financial support from the Czech Science Foundation (project No. 24-15057L). We would like to thank Michal Šámal from UOCHB for the synthesis of Helicene samples.