Effect of quartz and nanosilica on the hydration of LC3 cement
S. Švarcová1, P. Bezdička1, L. Scheinherrová2, M. Breníková2, M. Keppert2
1Institute of inorganic chemistry of the Czech academy of sciences v.v.i., Husinec-Řež 1001, 250 68 Husinec-Řež
2Department of materials engineering and chemistry, Faculty of civil engineering, Czech technical university in Prague, Thákurova 7, 166 29 Praha 6
svarcova@iic.cas.cz
Limestone calcined clay cements (LC3) are supposed to be perspective low-carbon alternative to conventional ordinary Portland cement (OPC) and blended cements. [1] LC3 cements typically contain 50 % (or less) of Portland clinker, 30 % of calcined clay (here metakaolin), 15 % of fine ground limestone and 5 % of gypsum. The reduced environmental impact lies obviously in the lower content of Portland clinker but contrary, the low content of clinker is responsible for the slower concrete strength gain which is highly unfavourable property in the current construction industry. Hence a way for acceleration of LC3 hydration and strengthening is searched. There is a lot of acceleration admixtures well established in OPC based concrete such as CaCl2, Ca(NO3)2 or water glass. [2] However, their applicability in LC3 systems may be limited compared to OPC.
The present research aims to evaluate the possible hydration acceleration in LC3 by means of fine grained quartz (Q) and amorphous nanosilica (nS). These admixtures were dosed in amount of 3 % by clinker mass to LC3 system of the common composition given above. The effect of addition of these admixtures was evaluated by isothermal calorimetry (20 °C), by determination of compressive strength in the age of 1 day (paste w/c 0.37, water reducing admixture was used to maintain proper workability) and finally by in situ XRPD obtained on the fresh paste, placed on PEEK foil, and measured in reflection mode in 1 hour period.
|
|
|
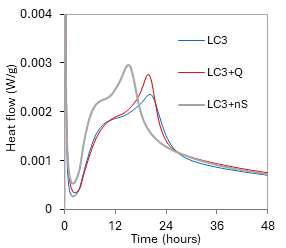
Figure 1. Hydration heat flow. |
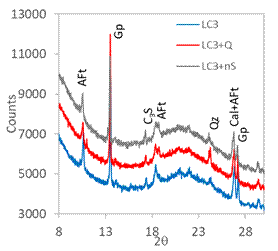
Figure 2. XRPD patterns in 4 hours. |
The shape of isothermal calorimetry heat flow curve of plain LC3 (Fig. 1) is very similar to conventional OPC systems; the strongly exothermic C3A hydration starts immediately after adding of water (time 0), then an induction (low heat flow) period appears and at about 3 hours, intensive heat evolution caused by C3S hydration starts. When quartz was added, the heat flow curve featured just a small change while nanosilica caused more pronounced increase of hydration heat as well as certain shortening of the induction period. This behaviour was confirmed also by the compressive strength measurement (Tab. 1) – quartz system reached the same value as plain LC3, while nanosilica caused significant increase of the 1-day strength.
Table 1. Compressive strength in 24 hours.
|
Sample |
LC3 |
LC3+Q |
LC+nS |
|
Compressive strength (MPa) |
4.5 |
4.7 |
7.9 |
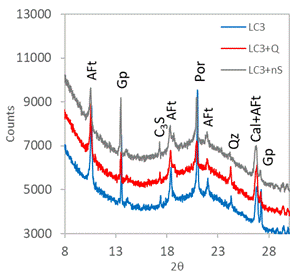
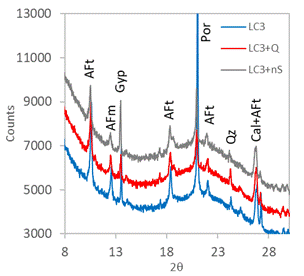
The in situ XRPD was used in order to explain how the quartz and nanosilica admixtures influence the hydration course. The XRPD patterns obtained in time of hydration 4 hours (initial part of main hydration peak; Fig. 2) are practically equal; the only observed crystalline hydration product in all systems was AFt phase (ettringite is the most prominent representative of AFt). Other observed phases come from the raw materials of LC3. After 12 hours (Fig. 3; intensive hydration heat flow) portlandite (Por, Ca(OH)2) appeared as another important hydration product. Portlandite is related to the C3S hydration. The intensity of portlandite diffraction at 21°2θ was the highest in the plain LC3, while the both admixtures reduced significantly its height. It implies that siliceous admixtures promoted the consumption of portlandite in the pozzolanic reaction with metakaolin, when compared with the plain LC3. There are also observable changes in gypsum (Gp) and AFt diffractions – SiO2 additives seems to reduce the AFt formation. The 24 hours data (end of the main hydration peak; Fig. 4) confirmed these trends, intensity of portlandite diffraction was again much higher in reference LC3 without additives. AFm phase (generic formula 3CaO·(Al,Fe)2O3·CaXy·nH2O) appeared at 24 h as product of AFt partial conversion (the peak at main hydration peak is supposed to be related to the AFm crystallization).
|
|
|
Figure 3. XRPD patterns in 12 hours. |
Figure 4. XRPD patterns in 24 hours. |
It can be concluded that both SiO2 admixtures promoted the pozzolanic reaction between Ca(OH)2 and metakaolin in LC3 but only nanosilica caused substantial increase of compressive strength. It may be due to the involvement of nanosilica in the pozzolanic reaction by itself (besides accelerating the reaction between Ca(OH)2 and metakaolin). The content of nanosilica is low compared to metakaolin but it may be assumed that it is much more reactive thank to its high specific surface area and low particle size.
1. K. Scrivener, F. Martirena, S. Bishnoi, S Maity, Cem. Conc. Res., 114, (2018), 49-56.
2. T. Dorn, O. Blask, D. Stephan, Con. Build. Mat., 323, (2022), 126554.
This work has been supported by Czech Science Foundation under project Nr. 25-15200S “Opportunities for increasing low early strength of eco-friendly blended cements through innovative additives and nanomaterials”.