Soft chemical control of the crystal structure and properties of Sr2MnO2Cu1.5-δS2
J. N. Blandy1, A. M. Abakumov2, K. E Christensen1, J. Hadermann2, P. Adamson1, D. G. Free1, H. Cohen1, A. L. Thompson1 and S. J. Clarke1
1Department of
Chemistry, University of Oxford, Inorganic Chemistry Laboratory, South Parks
Road, Oxford, OX1 3QR
2Electron Microscopy for Materials Science (EMAT), University of
Antwerp, Grenenborgerlaan 171, B-2020 Antwerp, Belgium
jack.blandy@chem.ox.ac.uk
Sr2MnO2Cu1.5-δS2[1] is a constituent compound of the wider class A2MO2X2Ch2 (A = Sr, Ba; M = 1st row transition metal, X = Cu, Ag; Ch = S, Se). Previous work on this compound has included investigation of its potential as a Li+ ion host in batteries [2] and examination of the solid solution Sr2MnO2Cu1.5S2-xSex.[3] This work explores topotactic oxidation of the manganese ions to synthesise metastable compositions with larger copper deficiencies.[4]
Sr2MnO2Cu1.5S2 was prepared via solid state synthesis, and then copper was deintercalated using varying concentrations of I2/MeCN solution. The high mobility of copper in this compound meant that a range of copper occupancies could be obtained between 1.5 and 1.33 with a phase gap in the region 1.39 – 1.35.
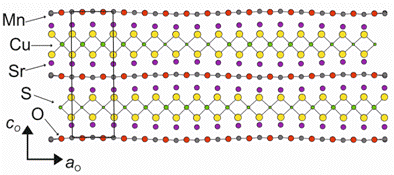
Reduction in copper content led to changes in the copper vacancy ordering pattern from that described by Adamson et al.[3], which only appears at temperatures below an ordering temperature of c. 250 K, to an incommensurately modulated ordering pattern, which is present in room temperature samples. This incommensurate ordering pattern was solved using a (3 + 1)D model approach, with modulation vector q = 0.2418 a0* and space group Xmmm(α00)00s. It was found that, as well as occupational modulation of the Cu atoms, there was also significant positional modulation of the oxide layers, which moved closer to the [Cu2-δS2] layers to ensure that the S atoms did not become underbonded in low Cu regions (fig. 1).
The presentation will also describe the effect of tuning composition and structure on physical properties.